Maxwell Boltzmann Distribution Law
Maxwell Boltzmann Distribution Law Assignment Help | Maxwell Boltzmann Distribution Law Homework Help
Maxwell-Boltzmann Distribution Law
Consider a system of N identical but distinguishable particles such as gas molecules at temperature T. Suppose the total volume of the system is V and total energy. Suppose n1 molecules are present in the level with energyand
Where n1 is the number of molecules in the ith level with energy
Many distributions are possible which satisfy the above two conditions. Now let us first see what the total number of such possible distributions is. The situation is similar to putting N numbered balls into n boxes such that n1are in the fist box, n2 in the second box and so on. Mathematically, the number of possible arrangement is given by
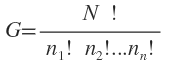
When applied to the system of N molecules, the number of possible distributions usually represented by W and is given by
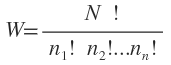
W is called the thermodynamic probability of the system because it is found that most of the thermodynamic properties are intimately related to the value of W ( e.g. S = k In W).
Now let us see ‘what is the most probable distribution’?
According to principles of statistical mechanics, the most probable distribution is one that makes W the maximum. This condition for maximum probability may be written mathematically in the form*
d = In W =0
Taking logarithm of both sides of equation we get
In
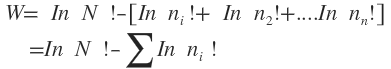 In
In Applying Sterling’s formula, according to which
and
eqn. is simplified to the form
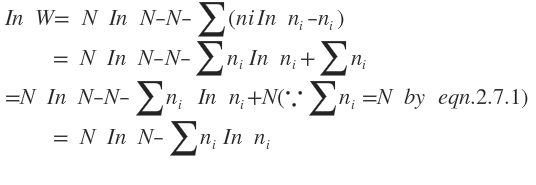
Differentiating both sides of this equating, we get
{d(N In N) = 0,Because N is constant}
(ni In ni has been differentiated as a product of two functions)
But d In W = 0 (according to eqn.)
Eqn. gives
Now as for the system under consideration
and
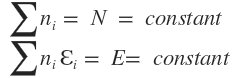
Therefore differentiation of these equations gives
and
Equations and (2.7.8), (2.7.9) and (2.7.10) represent the conditions which must be satisfied simultaneously for the most probable distribution.
Multiplying equations (2.7.9) and (2.7.10) by the undetermined constants
Putting
Now within the restrictions imposed on the system for constant N and E values, the variation in ( ) are independent of each other and need not be zero, i.e., ( ) . Consequently, equation (2.7.12) will be satisfied only If the term in the summating is zero. Thus
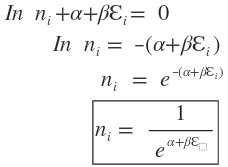
This expression is called Maxwell-Boltzmann distribution law
For more help in Maxwell-Boltzmann Distribution Law click the button below to submit your homework assignment